| Citation: | Jun Peng, Xianhui Yi, Ling Fan, Jiang Zhou, Bingan Lu. Molecular extension engineering constructing long-chain organic elastomeric interphase towards stable potassium storage[J]. Energy Lab, 2023, 1(2): 220014. doi: 10.54227/elab.20220014 |
Molecular extension engineering constructing long-chain organic elastomeric interphase towards stable potassium storage
-
Abstract
Electrolytes are critical for achieving high performance potassium ion batteries (PIBs) because of their ability to modulate the solid electrolyte interphase (SEI). However, the compositions of SEI in conventional electrolytes are either anion-derived inorganic-rich compounds or solvent molecule-derived short-chain organic-rich compounds. These SEI are generally inelastic and cannot effectively relieve the stress changes caused by volume changes during the charge/discharge processes. Here, we constructed long-chain organic-rich SEI (LO-SEI) with high elasticity by introducing a green and harmless long-chain solvent of dicaprylyl carbonate (DCC), thus greatly improving the performance of PIBs. As a result, a long stability of more than 1500 cycles (86.7% capacity retention) for graphite half-cells and more than 3700 hours for K||K symmetric cells are achieved. In addition, the elastomeric LO-SEI-based full cell is capable of stable operating for more than 130 cycles (84.3% capacity retention). This work may open new ideas for constructing long-chain elastic interphases to achieve high-performance batteries.
-
-
References
1. W. Zhang, Y. Liu, Z. Guo, Sci. Adv., 2019, 5, eaav7412 2. J. Li, Y. Hu, H. Xie, J. Peng, L. Fan, J. Zhou, B. Lu, Angew. Chem. Int. Ed., 2022, 61, e202208291 3. B. Xiao, H. Zhang, Z. Sun, M. Li, Y. Fan, H. Lin, H. Liu, B. Jiang, Y. Shen, M.-S. Wang, M. Li, Q. Zhang, J. Energy Chem., 2023, 76, 547 4. M. Ma, S. Zhang, L. Wang, Y. Yao, R. Shao, L. Shen, L. Yu, J. Dai, Y. Jiang, X. Cheng, Y. Wu, X. Wu, X. Yao, Q. Zhang, Y. Yu, Adv. Mater., 2021, 33, 2106232 5. L. Fan, R. Ma, Q. Zhang, X. Jia, B. Lu, Angew. Chem. Int. Ed., 2019, 58, 10500 6. Y. Hu, L. Fan, A. M. Rao, W. Yu, C. Zhuoma, Y. Feng, Z. Qin, J. Zhou, B. Lu, Natl. Sci. Rev., 2022, 9, nwac134 7. L. A. Schkeryantz, J. Zheng, W. D. McCulloch, L. Qin, S. Zhang, C. E. Moore, Y. Wu, Chem. Mater., 2020, 32, 10423 8. Z. Yu, P. E. Rudnicki, Z. Zhang, Z. Huang, H. Celik, S. T. Oyakhire, Y. Chen, X. Kong, S. C. Kim, X. Xiao, H. Wang, Y. Zheng, G. A. Kamat, M. S. Kim, S. F. Bent, J. Qin, Y. Cui, Z. Bao, Nat. Energy, 2022, 7, 94 9. S. Liu, J. Mao, Q. Zhang, Z. Wang, W. K. Pang, L. Zhang, A. Du, V. Sencadas, W. Zhang, Z. Guo, Angew. Chem. Int. Ed., 2020, 59, 3638 10. J. Ming, Z. Cao, W. Wahyudi, M. Li, P. Kumar, Y. Wu, J.-Y. Hwang, M. N. Hedhili, L. Cavallo, Y.-K. Sun, L.-J. Li, ACS Energy Lett., 2018, 3, 335 11. E. P. Kamphaus, S. Angarita-Gomez, X. Qin, M. Shao, M. Engelhard, K. T. Mueller, V. Murugesan, P. B. Balbuena, ACS Appl. Mater. Interfaces, 2019, 11, 31467 12. H. Wang, D. Zhai, F. Kang, Energy Environ. Sci., 2020, 13, 4583 13. M. Gauthier, T. J. Carney, A. Grimaud, L. Giordano, N. Pour, H.-H. Chang, D. P. Fenning, S. F. Lux, O. Paschos, C. Bauer, F. Maglia, S. Lupart, P. Lamp, Y. Shao-Horn, J. Phys. Chem. Lett., 2015, 6, 4653 14. Y. Lei, D. Han, J. Dong, L. Qin, X. Li, D. Zhai, B. Li, Y. Wu, F. Kang, Energy Stor. Mater., 2020, 24, 319 15. Y. Li, Q. Zhang, Y. Yuan, H. Liu, C. Yang, Z. Lin, J. Lu, Adv. Energy Mater., 2020, 10, 2000717 16. K. Shi, L. Chen, Z. Wan, J. Biao, G. Zhong, X. Li, L. Yang, J. Ma, W. Lv, F. Ren, H. wang, Y. Yang, F. Kang, Y.-B. He, Sci. Bull., 2022, 67, 946 17. Z. Chen, B. Wang, Y. Li, F. Bai, Y. Zhou, C. Li, T. Li, ACS Appl. Mater. Interfaces, 2022, 14, 28014 18. X. Zheng, L. Huang, W. Luo, H. Wang, Y. Dai, X. Liu, Z. Wang, H. Zheng, Y. Huang, ACS Energy Lett., 2021, 6, 2054 19. Z. Hou, R. Zhou, Y. Yao, Z. Min, Z. Lu, Y. Zhu, J.-M. Tarascon, B. Zhang, Angew Chem. Int. Ed., 2022, 61, e202214796 20. B. Gangaja, S. Nair, D. Santhanagopalan, Sustain. Energy Fuels, 2019, 3, 2490 21. S. Liu, X. Ji, N. Piao, J. Chen, N. Eidson, J. Xu, P. Wang, L. Chen, J. Zhang, T. Deng, S. Hou, T. Jin, H. Wan, J. Li, J. Tu, C. Wang, Angew. Chem. Int. Ed., 2021, 60, 3661 22. L. Droguet, G. M. Hobold, M. F. Lagadec, R. Guo, C. Lethien, M. Hallot, O. Fontaine, J.-M. Tarascon, B. M. Gallant, A. Grimaud, ACS Energy Lett., 2021, 6, 2575 23. X. Fan, X. Ji, F. Han, J. Yue, J. Chen, L. Chen, T. Deng, J. Jiang, C. Wang, Sci. Adv., 2018, 4, eaau9245 24. K. Subramanian, G. V. Alexander, K. Karthik, S. Patra, M. S. Indu, O. V. Sreejith, R. Viswanathan, J. Narayanasamy, R. Murugan, J. Energy Stor., 2021, 33, 102157 25. C. Fang, J. Li, M. Zhang, Y. Zhang, F. Yang, J. Z. Lee, M.-H. Lee, J. Alvarado, M. A. Schroeder, Y. Yang, B. Lu, N. Williams, M. Ceja, L. Yang, M. Cai, J. Gu, K. Xu, X. Wang, Y. S. Meng, Nature, 2019, 572, 511 26. H. Kitaura, E. Hosono, H. Zhou, Energy Environ. Sci, 2021, 14, 4474 27. A. Ramasubramanian, V. Yurkiv, T. Foroozan, M. Ragone, R. Shahbazian-Yassar, F. Mashayek, J. Phys. Chem. C, 2019, 123, 10237 28. J. Pan, Q. Zhang, X. Xiao, Y.-T. Cheng, Y. Qi, ACS Appl. Mater. Interfaces, 2016, 8, 5687 29. S. Yuan, S. Weng, F. Wang, X. Dong, Y. Wang, Z. Wang, C. Shen, J. L. Bao, X. Wang, Y. Xia, Nano Energy, 2021, 83, 105847 30. C. Stetson, M. Schnabel, Z. Li, S. P. Harvey, C.-S. Jiang, A. Norman, S. C. DeCaluwe, M. Al-Jassim, A. Burrell, ACS Energy Lett., 2020, 5, 3657 31. Y. Rangom, R. R. Gaddam, T. T. Duignan, X. S. Zhao, ACS Appl. Mater. Interfaces, 2019, 11, 34796 32. J. Zhu, P. Li, X. Chen, D. Legut, Y. Fan, R. Zhang, Y. Lu, X. Cheng, Q. Zhang, Energy Stor. Mater., 2019, 16, 426 33. F. Yuan, J. Hu, Y. Lei, R. Zhao, C. Gao, H. Wang, B. Li, F. Kang, D. Zhai, ACS Nano, 2022, 16, 12511 34. K. Yan, H.-W. Lee, T. Gao, G. Zheng, H. Yao, H. Wang, Z. Lu, Y. Zhou, Z. Liang, Z. Liu, S. Chu, Y. Cui, Nano Lett., 2014, 14, 6016 35. J. Fondard, E. Irisarri, C. Courrèges, M. R. Palacin, A. Ponrouch, R. Dedryvère, J. Electrochem. Soc., 2020, 167, 070526 36. L. Ma, C. Fu, L. Li, K. S. Mayilvahanan, T. Watkins, B. R. Perdue, K. R. Zavadil, B. A. Helms, Nano Lett., 2019, 19, 1387 37. Y. Zhao, M. Amirmaleki, Q. Sun, C. Zhao, A. Codirenzi, L. V. Goncharova, C. Wang, K. Adair, X. Li, X. Yang, F. Zhao, R. Li, T. Filleter, M. Cai, X. Sun, Matter, 2019, 1, 1215 38. Y. Zhou, M. Su, X. Yu, Y. Zhang, J.-G. Wang, X. Ren, R. Cao, W. Xu, D. R. Baer, Y. Du, O. Borodin, Y. Wang, X.-L. Wang, K. Xu, Z. Xu, C. Wang, Z. Zhu, Nat. Nanotechnol., 2020, 15, 224 39. Z. Wang, C. Sun, Y. Shi, F. Qi, Q. Wei, X. Li, Z. Sun, B. An, F. Li, J. Power Sources, 2019, 439, 227073 40. D. Kang, S. Sardar, R. Zhang, H. Noam, J. Chen, L. Ma, W. Liang, C. Shi, J. P. Lemmon, Energy Stor. Mater., 2020, 27, 69 41. X.-Q. Zhang, X.-B. Cheng, X. Chen, C. Yan, Q. Zhang, Adv. Funct. Mater., 2017, 27, 1605989 42. X. Ren, Y. Zhang, M. H. Engelhard, Q. Li, J.-G. Zhang, W. Xu, ACS Energy Lett., 2018, 3, 14 43. Y. Liu, X. Tao, Y. Wang, C. Jiang, C. Ma, O. Sheng, G. Lu, X. Wen, Science, 2022, 375, 739 44. Z. Lin, X. Guo, R. Zhang, M. Tang, P. Ding, Z. Zhang, L. Wu, Y. Wang, S. Zhao, Q. Zhang, H. Yu, Nano Energy, 2022, 98, 107330 45. Q. Liu, A. M. Rao, X. Han, B. Lu, Adv. Sci., 2021, 8, 2003639 46. D. Hulicova-Jurcakova, M. Kodama, S. Shiraishi, H. Hatori, Z. H. Zhu, G. Q. Lu, Adv. Funct. Mater., 2009, 19, 1800 47. Z. Gu, G. Li, N. Hussain, B. Tian, Y. Shi, Appl. Surface Sci., 2022, 592, 153323 48. A. Ponrouch, R. Dedryvère, D. Monti, A. E. Demet, J. M. Ateba Mba, L. Croguennec, C. Masquelier, P. Johansson, M. R. Palacín, Energy Environ. Sci., 2013, 6, 2361 49. Y. Gao, X. Du, Z. Hou, X. Shen, Y.-W. Mai, J.-M. Tarascon, B. Zhang, Joule, 2021, 5, 1860 50. X.-Q. Zhang, X. Chen, X.-B. Cheng, B.-Q. Li, X. Shen, C. Yan, J.-Q. Huang, Q. Zhang, Angew. Chem. Int. Ed., 2018, 57, 5301 51. D. Lu, X. Lei, S. Weng, R. Li, J. Li, L. Lv, H. Zhang, Y. Huang, J. Zhang, S. Zhang, L. Fan, X. Wang, L. Chen, G. Cui, D. Su, X. Fan, Energy Environ. Sci., 2022, 15, 3331 -
Rights and permissions
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Information
Article Metrics
-
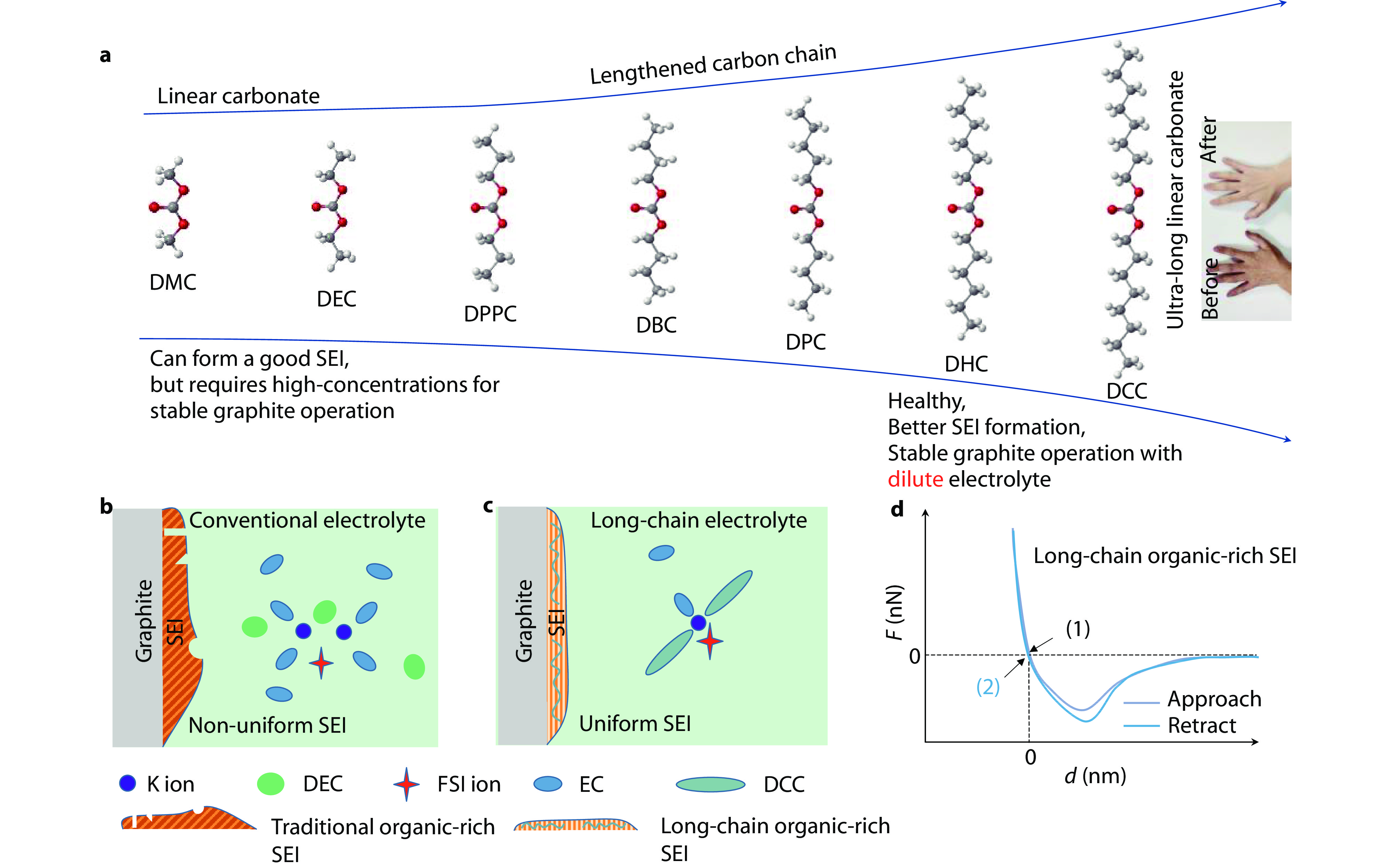
Figure 1.
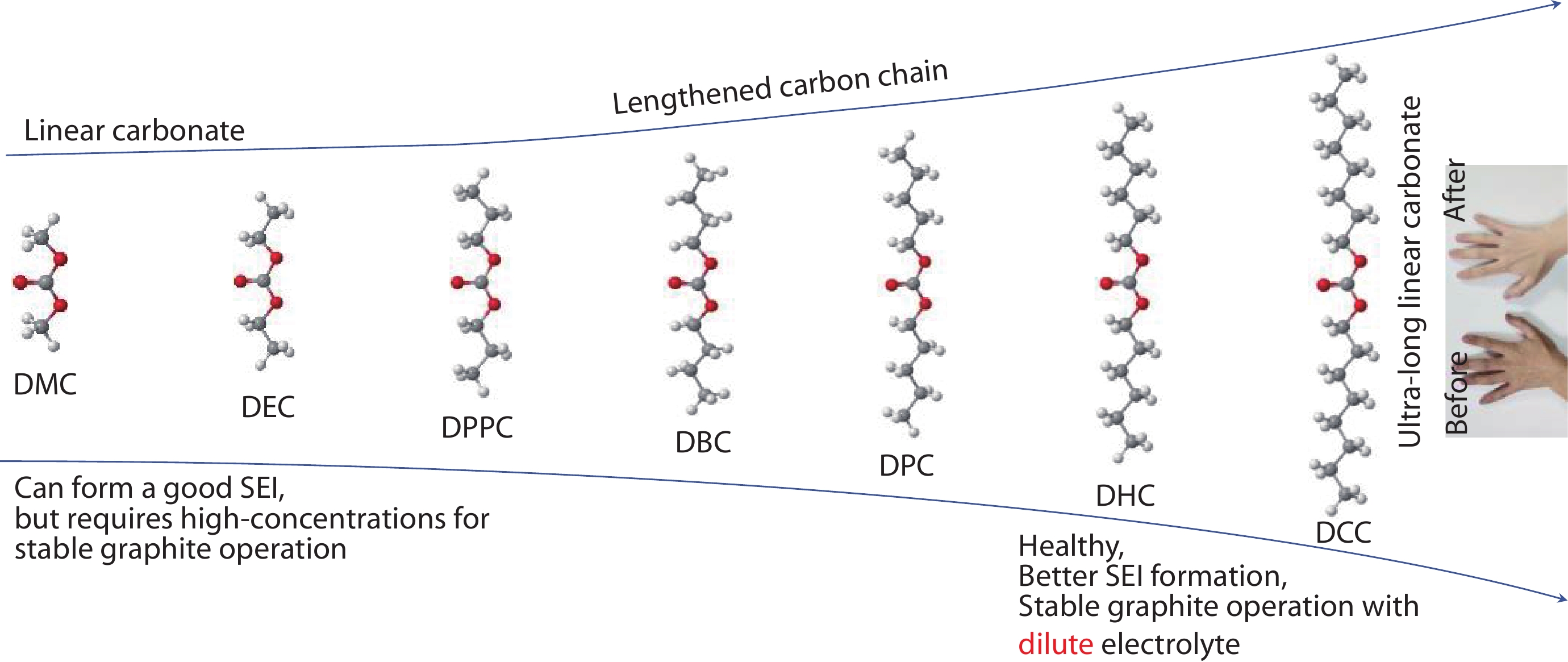
a Molecular design to grow a range of products with terminal substituents and to regulate the solvation properties of solvent molecules. The small picture shows the comparison before and after applying the hand with DCC solvent. SEI morphology on graphite anode and solvation structure of b conventional and c long-chain electrolytes. d Schematic diagram of the effect of long-chain organic molecules on the elasticity of SEI.
-
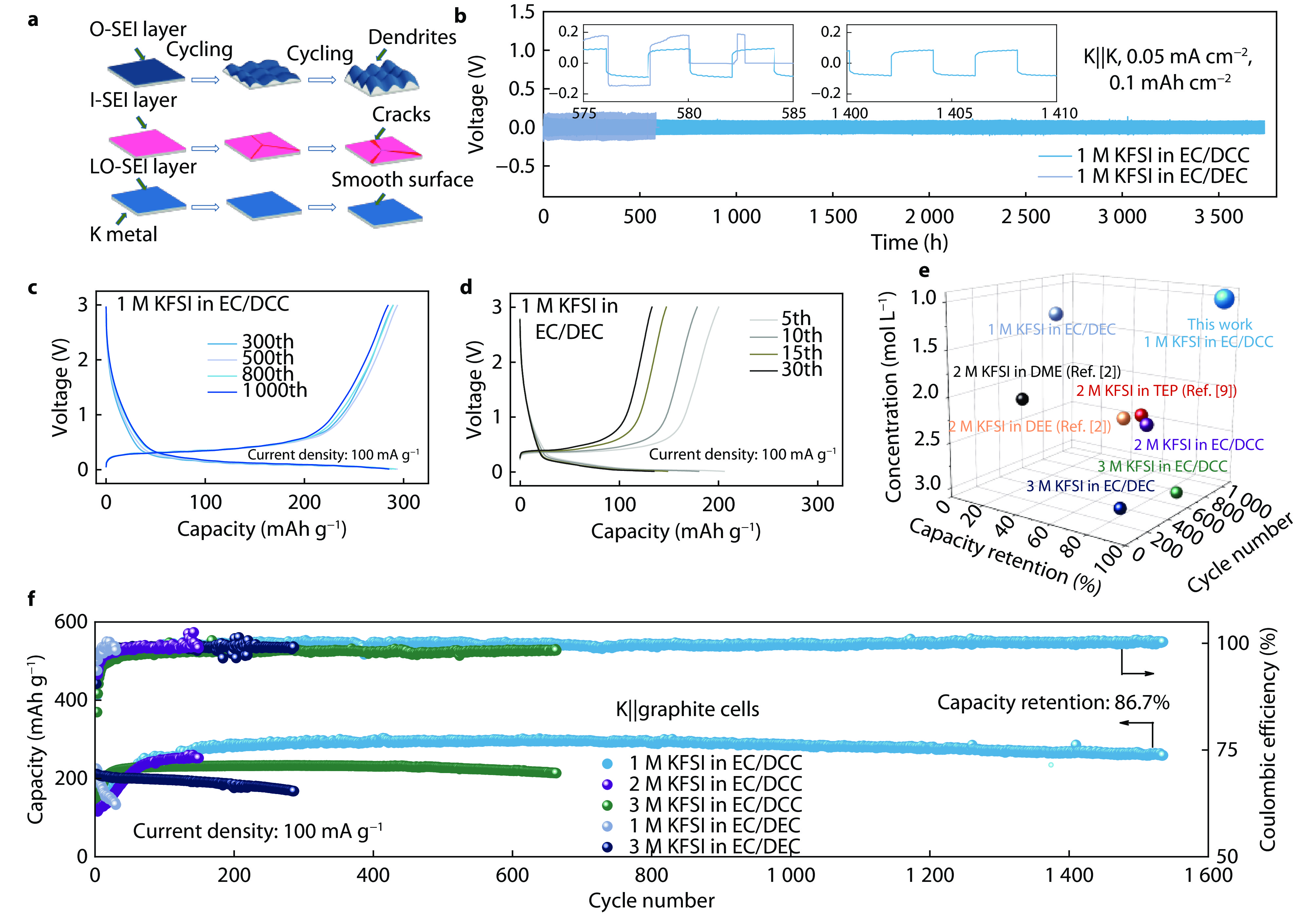
Figure 2.
a The schematic diagram of three kinds of SEI for K metal anode. b Comparison of the performance of 1 M KFSI in EC/DEC and 1 M KFSI in EC/DCC electrolytes for symmetric K||K cells at a current density of 0.05 mA cm−2. Charge/discharge voltage profile of graphite anode in c 1 M KFSI in EC/DCC and d 1 M KFSI in EC/DEC electrolytes. e Comparison of cycling performance, concentration and capacity retention between graphite electrodes with different electrolytes cycles (TEP is triethyl phosphate, DME means 1,2-dimethoxyethane and DEE stands for 1,2-diethoxyethane). f Cycling performance of K||graphite cells with Coulombic efficiency with five different electrolytes at a current density of 100 mA g−1.
-
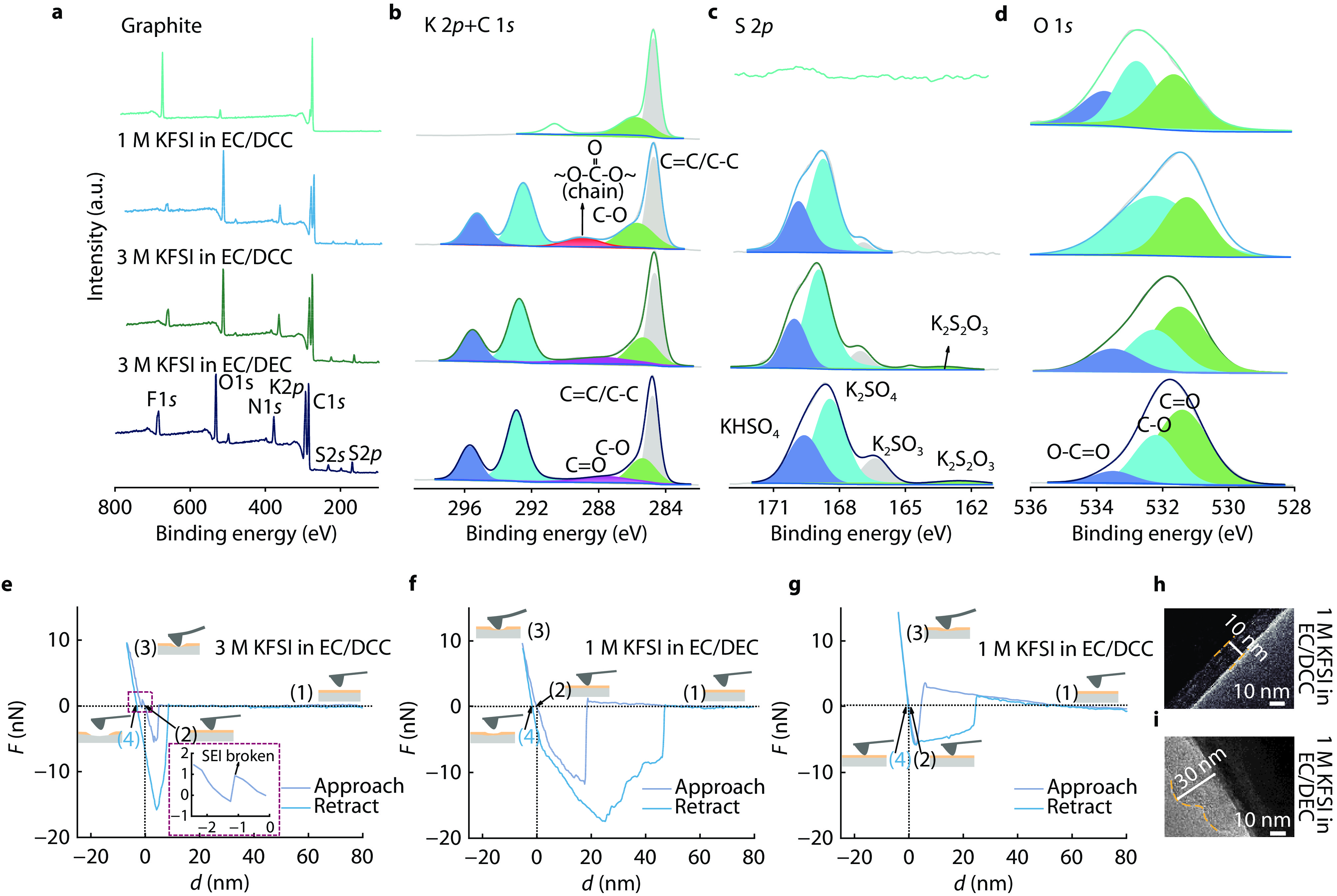
Figure 3.
XPS analysis of graphite electrode after 5 cycles with initial graphite and three kinds of electrolytes (1 M KFSI in EC/DCC, 1 M KFSI in EC/DEC, 3 M KFSI in EC/DEC). a Full survey XPS. b K 2p + C 1s XPS. c S 2p XPS. d O 1s XPS. Characterizations of the SEI formed on graphite electrodes with two different electrolytes after 20 cycles. Graphite after 10 cycles for AFM stress test of e 3 M KFSI in EC/DCC, f 1 M KFSI in EC/DEC and g 1 M KFSI in EC/DCC. TEM images of graphite cathodes after 10 cycles in h 1 M KFSI in EC/DCC and i 1 M KFSI in EC/DEC electrolytes. The yellow dashed lines indicate partial SEI.
-
Figure 4.
a Charge/discharge voltage profile of PTCDI cathode in 1 M KFSI in EC/DCC electrolyte. b Cycling performance of K||PTCDI cell at a current density of 100 mA g−1. c Normalized charge/discharge profiles of full and half cells based on graphite anodes and PTCDI cathodes. d Charge/discharge profiles of different cycles and lighted signs lit by full batteries. e Cycling performance of a full cell at 80 mA g−1. f Comparison of the properties and performances of the 1 M KFSI in EC/DCC and EC/DEC electrolytes.


 DownLoad:
DownLoad: